Class 10 Science Chapter 5 Important Questions Periodic Classification of Elements
Class 10 Science Chapter 5 Important Questions Periodic Classification of Elements, (Science) exams are Students are taught thru NCERT books in some of the state board and CBSE Schools. As the chapter involves an end, there is an exercise provided to assist students to prepare for evaluation. Students need to clear up those exercises very well because the questions inside the very last asked from those.
Sometimes, students get stuck inside the exercises and are not able to clear up all of the questions. To assist students, solve all of the questions, and maintain their studies without a doubt, we have provided a step-by-step NCERT Important Questions for the students for all classes. These answers will similarly help students in scoring better marks with the assist of properly illustrated Notes as a way to similarly assist the students and answer the questions right.
Class 10 Science Chapter 5 Important Questions Periodic Classification of Elements
Objective type questions
Stand Alone MCQS (1 Mark Each)
1. The positions of four elements A, B, Cand D in the modern periodic table are shown below. Which element is most likely to form an acidic oxide?
(A) A
(B) B
(C) C
(D) D
Ans. Option (C) is correct.
Explanation: Element C is most likely to form an acidic oxide because it lies in non-metal category.
2. Elements P, Q, R and S have atomic numbers 11, 15, 17 and 18 respectively. Which of them are reactive non-metals?
(A) P and Q
(B) P and R
(C) Q and R
(D) R and S
Ans. Option (C) is correct.
Explanation: Element Q and R are reactive non-metals because they have great tendency to accept the electrons to occupy the stable electronic configuration.
3. Consider the following statements about an element ‘X’ with number of protons 13.
(I) It forms amphoteric oxide.
(II) Its valency is three.
(III) The formula of its chloride is XCI.
(A) only (I)
(B) only (II)
(C) (I) and (III)
(D) (I) and (II)
Ans. Option (D) is correct.
Explanation: The element is Al. It forms amphoteric oxide Al2O3, its valency is three and the formula of chloride is AICL3.
4. Upto which element, the Law of Octaves was found to be applicable?
(A) Oxygen
(B) Calcium
(C) Cobalt
(D) Potassium
Ans. Option (B) is correct.
Explanation: It was found that the Law of Octaves was applicable only upto calcium, as after calcium every eighth element did not possess properties similar to that of the first.
5. According to Mendeleev’s Periodic Law, the elements were arranged in the periodic table in the order of
(A) increasing atomic number
(B) decreasing atomic number
(C) increasing atomic masses
(D) decreasing atomic masses
Ans. Option (C) is correct.
Explanation: Mendeleev arranged the known elements according to increasing order of their atomic masses because according to him, fundamental property of an element was atomic mass.
6. In Mendeleev’s periodic table, gaps were left for the elements to be discovered later. Which of the following elements found a place in the periodic table later?
(A) Germanium
(B) Chlorine
(C) Oxygen
(D) Silicon
Ans. Option (A) is correct.
Explanation:
- Many gaps for the undiscovered elements were left in the periodic table by Mendeleev.
- For instance, the elements scandium (Sc), gallium (Ga), and garmanium (Ge) were not known at Mendeleev’s time, but he had predicted their existence in advance of their discovery.
7. What type of oxide would Eka- aluminium form?
(A) EO3
(B) E2O2
(C) E2O3
(D) EO
Ans. Option (C) is correct.
Explanation:
- Eka-aluminium belongs to group 13.
- Hence, formula of oxide in group 13 is E2O3.
8. Which of these does not represent Dobereiner’s triad?
(A) Li, Na, K
(B) CI, Br, I
(C) Be, Mg, Ca
(D) N, P, As
Ans. Option (D) is correct.
Explanation:
- N, P and as will not represent Dobereiner’s triad.
- It is because; the average of atomic masses of Nitrogen and Arsenic is not equal to atomic mass of phosphorus.
9. An element ‘X is forming an acidic oxide. Its position in modern periodic table will be:
(A) Group 1 and Period 3
(B) Group 2 and Period 3 U
(C) Group 13 and Period 3
(D) Group 16 and Period 3
Ans. Option (D) is correct.
Explanation: Elements of Group 16 and Period 3 are non-metals that form acidic oxides.
10. Where would you locate the element with electronic configuration 2, 8 in the Modern Periodic Table?
(A) Group 8
(B) Group 2
(C) Group 18
Ans. Option (C) is correct.
Explanation: Element with electronic configuration 2, 8 has complete octet so must be placed in group 18.
11. Which of the following is the outermost shell for elements of period 2?
(A) K shell
(B) L shell
(C) M shell
(D) N shell
Ans. Option (B) is correct.
Explanation: The elements of 2nd period contain two shells, K and L shell.
12. Arrange the following elements in the order of their decreasing metallic character: Na, Si, CI, Mg, Al.
(A) Cl > Si >Al > Mg >Na
(B) Na >Mg >Al >Si > Cl
(C) Na > Al > Mg > Cl > Si
(D) Al > Na> Si > Ca> Mg
Ans. Option (B) is correct.
Explanation: As we move from left to right in periodic table, metallic characters decreases, and non-metallic characters increases.
13. Which of the following elements will form an acidic oxide?
(A) An element with atomic number 7
(B) An element with atomic number 3
(C) An element with atomic number 12
(D) An element with atomic number 19
Ans. Option (A) is correct.
Explanation:
- Formation of acidic oxides is a characteristic of non-metals.
- Here, element with atomic number 7 is a non-metal i.e., nitrogen.
- Rest three elements are metals and hence metals form basic oxide.
14. Which of the following set of elements is written in order of their increasing metallic character?
(A) Be, Mg, Ca
(C) Mg, Al, Si
(B) Na, Li, K
(D) C, O, N
Ans. Option (A) is correct.
Explanation:
- As we move down in a group, metallic nature increases due to increase in electron losing tendency.
- Be, Mg and Ca belong to same group i.e., Group 2 of Modern Periodic table.
15. Which of the following are the characteristics of isotopes of an element?
(i) Isotopes of an element have same atomic masses.
(ii) Isotopes of an element have same atomic number.
(iii) Isotopes of an element show same physical properties.
(iv) Isotopes of an element show same chemical properties.
(A) (i), (iii) and (iv)
(B) (ii), (iii) and (iv)
(C) (ii) and (iii)
(D) (ii) and (iv)
Ans. Option (D) is correct.
Explanation:
- Isotopes are the atoms of the same element which have same atomic number because of same number of valence electrons but different number of neutrons hence their atomic masses are different.
- This leads to similar chemical properties but different physical properties.
16. Which of the following statements is not a correct statement about the trends when going from left to right across the periods of periodic table?
(A) The elements become less metallic in nature
(B) The number of valence electrons increases
(C) The atoms lose their electrons more easily
(D) The oxides become more acidic.
Ans. Option (C) is correct.
Explanation:
- On moving from left to right across the periods of the periodic table, the non-metallic character increases.
- Hence, the tendency to lose electrons decreases.
Assertion and Reason Based MCQS (1 Mark Each)
Directions: In the following questions, A statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as.
(A) Both A and R are true, and R is the correct explanation of A.
(B) Both A and R are true, but R is NOT the correct explanation of A.
(C) A is true but R is false.
(D) A is false and R is true.
1. Assertion (A): Mendeleev arranged element in horizontal rows and vertical columns.
Reason (R): Mendeleev ignored the order of atomic weight thinking that the atomic measurements might be incorrect.
Ans. Option (A) is correct.
Explanation:
- Mendeleev arranged elements in horizontal rows and vertical columns.
- He ignored the order of atomic weight thinking that the atomic measurements might be incorrect and placed the elements with similar properties.
2. Assertion (A): Mendeleev left the gaps under aluminium Eka-aluminium and Eka-silicon, respectively.
Reason (R): Dobereiner arranged elements on the basis of increasing atomic number.
Ans. Option (C) is correct.
Explanation: Dobereiner arranged elements on the basis of increasing atomic weights in a triad.
3. Assertion (A): In a triad, the three elements have same gaps between their atomic masses.
Reason (R): Elements in a triad have similar properties.
Ans. Option (D) is correct.
Explanation:
- In a triad, the atomic mass of the middle element is the mean of the atomic masses of the first and third elements.
- These elements have similar physical and chemical properties.
4. Assertion (A): According to Mendeleev, periodic properties of elements is a function of their atomic number.
Reason (R): Atomic number is equal to the number of protons.
Ans. Option (D) is correct.
Explanation:
- According to Mendeleev, periodic properties of elements is a function of their atomic masses.
- Atomic number is defined by the number of protons present in the nucleus of atoms of the element.
5. Assertion (A): Sixth and seventh periods in the periodic table contains 14 elements.
Reason (R): In the periodic table, 14 elements of sixth and seventh periods are known as lanthanoids and actinoids respectively.
Ans. Option (D) is correct.
Explanation:
- Sixth and seventh period contains 32 elements.
- Out of 32 elements 14 are the lanthanoids in period 6 and actinoids in period 7.
6. Assertion (A): Be and Al show similar properties.
Reason (R): The metallic radius of Be is less than the metallic radius of Al.
Ans. Option (B) is correct.
Explanation:
- Be and Al show diagonal relationship because Be resembles in its properties with Al.
- Metallic radius of the Be (111 pm) is less than the metallic radius of Al (143 pm).
- Although smaller size is the reason for the anomalous behaviour of Be but not a reason for its diagonal relation with Al.
7. Assertion (A): The atomic and ionic radii generally decrease towards right in a period.
Reason (R): The ionisation enthalpy increases on moving towards left in a period.
Ans. Option (C) is correct.
Explanation:
The ionisation enthalpy increases on moving towards the extreme right element in period due to increase in effective nuclear charge and atomic and ionic radii decreases in a period from left to right.
8. Assertion (A): Smaller the size of an atom, greater is the electronegativity.
Reason (R): Electronegativity refers to the tendency of atom to share electrons with another atom.
Ans. Option (C) is correct.
Explanation:
- Electronegativity refers to the tendency of an atom to attract shared pair of electrons toward itself.
- Down the group electronegativity decrease with increase in size.
9. Assertion (A): Noble gases are highly reactive.
Reason (R): Noble gases have stable closed shell electronic configuration.
Ans. Option (D) is correct.
Explanation: Inert gases (noble gases) are least reactive due to stable closed shell electronic configuration like 1s2 or ns2, np6 (Here n ≤ 2)
Case-based MCQS (1 Mark Each)
I. Read the passage and answer the questions given below:
Russian chemist, who Mendeleev contributed the most for the development of periodic table of elements wherein the elements were arranged on the basis of their fundamental property, the atomic mass and also on the similarity of chemical properties. Only 63 elements were known at his time. He arranged the 63 elements in the increasing order of their atomic masses and found that there was a periodic recurrence of elements with similar physical and chemical properties. He observed that elements with similar properties fall in the same vertical column. These vertical columns are called groups and horizontal rows of elements are called periods. Mendeleev predicted the existence of certain elements not known at that time and named two of them as Eka-silicon and Eka-aluminium was
1. Mendeleev arranged the periodic table on the basis of which fundamental property?
(A) Atomic mass
(B) Atomic number
(C) Number of neutrons
(D) Valence electrons
Ans. Option (A) is correct.
Explanation: Mendeleev arranged the known elements according to increasing order of their atomic masses because according to him, fundamental property of an element was atomic mass.
2. Eka aluminium and eka silicon were later replaced respectively as:
(A) Germanium and gallium
(B) Gallium and scandium
(C) Gallium and germanium
(D) Germanium and scandium
Ans. Option (C) is correct.
Explanation: Mendeleev defined unnamed elements as eka- Boron, eka- Aluminium and eka- Silicon which were later replaced as Scandium, Gallium, and Germanium respectively.
3. The elements eka aluminium and eka silicon discovered by Mendeleev later found place in periodic table. Both of these elements belong to:
(A) Period 2
(B) Group 13
(C) Group 14
(D) Period 4
Ans. Option (D) is correct.
Explanation: Eka silicon replaced with Germanium: Group 14, Period 4 and Eka aluminium replaced with Gallium: Group 13, Period 4.
4. How do we classify these elements (eka-aluminium and eka-silicon)?
(A) Metals
(B) Nonmetals
(C) Metalloids
(D) Inert gases
Ans. Option (C) is correct.
Explanation:
- The newly discovered elements were metalloids.
- A metalloid is an element that has properties that are intermediate between those of metals and non-metals.
II. Using the given part of the periodic table, answer the questions given below:
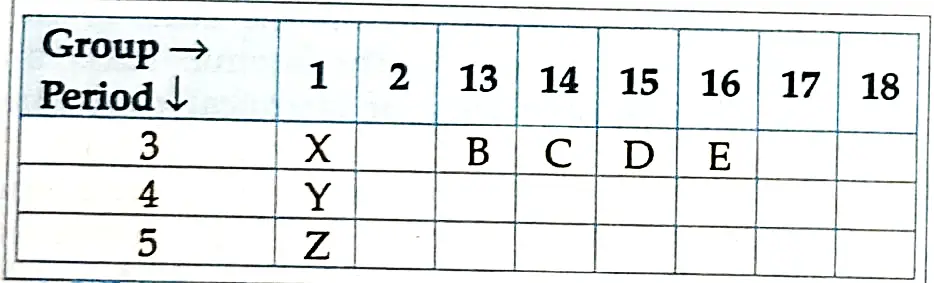
1. Which of these elements have smallest atomic size?
(A) B
(B) C
(C) D
(D) E
Ans. Option (D) is correct.
Explanation: Element E has the smallest atomic size because moving left to right of a period, atomic size decreases.
2. What is the valency of element E?
(A) 1
(B) 3
(C) 2
(D) 0
Ans. Option (C) is correct.
Explanation: The valency of element E is 2 as E belong to Group 16 and valency in Group 16 is always 2.
3. Identify the elements which have similar chemical properties as the element X.
(A) Y and Z.
(B) Y and B
(C) All Y, Z and B
(D) None of these
Ans. Option (A) is correct.
Explanation: Y and Z have similar physical and chemical properties as X.
4. Which of them will have largest atomic radii?
(A) E
(B) X
(C) C
(D) D
Ans. Option (B) is correct.
Explanation: X has the largest atomic radii. It is because atomic radii decrease from left to right along a period.
III. Read the passage and answer the questions given below:
Atoms of eight elements A, B, C, D, E, F, G and H have the same number of electronic shells as K, L and M. But are different in their outermost shells. It was found that elements A and G combine to form an ionic compound which can also be ex- tracted from sea water. Oxides of the elements A and B are basic in nature while those of E and F are acidic. The oxide of elements D is almost neu- tral.
1. To which period the listed elements belong?
(A) 2nd
(B) 7th
(C) 8th
(D) 3rd
Ans. Option (D) is correct.
Explanation:
- The eight elements belong to the 3rd period as they have the same number of electronic shells i.e., K, L and M but different number of electrons in their outermost shells.
- The number of valence electrons in these elements increases from 1 to 8, on moving from left to right in this period.
2. Which two of these elements could definitely be metals?
(A) E and F
(B) A and B
(C) D
(D) G
Ans. Option (B) is correct.
Explanation: A, B and C are definitely metals because in the 3rd period, at the extreme left, only metals are placed whose oxides are basic, D is a metalloid that have amphoteric oxides while E, F and G are non-metals that have acidic oxide.
3. Which two elements amongst these are likely to be the non-metals?
(A) A and G
(B) D and F
(C) E and F
(D) A and B
Ans. Option (C) is correct.
Explanation: E and F belong to group 15 and 16 as they form acidic oxides.
4. Which one of the following is most likely to be found in gaseous state at room temperature?
(A) A
(B) H
(C) D
(D) B
Ans. Option (B) is correct.
Explanation:
- The element H is most likely to be found in gaseous state at room temperature.
- It is because the last group elements in the periodic table are all in a gaseous state at room temperature.
IV. From the following table of the periodic table, answer the questions given:

1. Which is the most reactive metal?
(A) Lithium
(B) X
(C) Y
(D) Z
Ans. Option (D) is correct.
Explanation: Z is the most reactive metal as moving down the group electrons can be losed easily.
2. Name the family of fluorine, Q, R, T:
(A) Alkali metals
(B) Noble gas
(C) Halogens
(D) Alkaline metals
Ans. Option (C) is correct.
Explanation: The family of fluorine Q, R and T are Halogen because they belong to 17th group.
3. Which of the following element belongs to group 2?
(A) Sodium
(B) Magnesium
(C) Aluminium
(D) Carbon
Ans. Option (B) is correct.
Explanation: Magnesium belong to group 2, Sodium, Carbon and aluminium belong to group 1, group 14, group 13 respectively.
4. Which other element is likely to present in the group in which fluorine is present?
(A) Neon
(B) Aluminium
(C) Chlorine
(D) None of the above
Ans. Option (C) is correct.
Explanation: The halogen elements fluorine (F), chlorine (CI), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).
Subjective type questions
Very Short Answer Type Questions (1 Mark Each)
1. How many horizontal rows are present in modern periodic table? What are they called?
Ans. There are seven horizontal rows in the modern periodic table. These rows are called periods.
2. How many vertical columns are present in modern periodic table? What are they called?
Ans. There are 18 vertical columns in the modern periodic table, and these are known as groups.
3. State the formula to fill up the maximum number of electrons in a shell.
Ans. 2n2, where n is the number of shell.
4. Write the total number of periods in modern periodic table.
Ans. The total number of periods in modern periodic table is seven.
5. State one reason that explains the position of Hydrogen in group I.
Ans. Hydrogen should be placed in group I, since it has only one electron in its outermost shell.
6. Why noble gases are placed in a separate group in modern periodic table?
Ans. It is because they resemble with each other but do not resemble with other group elements.
7. How does valency of an element vary across a period?
Ans. The valency of an element first increases and then decreases across a period.
8. Define electropositivity.
Ans. Electropositivity is the tendency of an element to lose electrons and form positive ions in a chemical reaction.
9. The atomic radii of first group elements are given below:
Group I element atomic radii (pm)
Na 86
K 231
Rb 244
Cs 282
State the reason behind the observed trend in the above elements.
Ans. In a group, as we move from top to bottom, the number of shells increases. Hence, the atomic radius increases.
10. State one reason for placing Mg and Ca in the same group of the periodic table.
Ans. Due to the presence of 2 electrons in the valence shell and similar chemical properties.
11. How many elements are in 2nd and 5th period of Modern Periodic table.
Ans. 2nd period has 8 elements, 5th period has 18 elements.
12. Name any two elements that have two electrons in their valence shell.
Ans. (i) Magnesium: 2, 8, 2 (ii) Calcium: 2, 8, 8, 2.
13. If the atonmic number of three element X, Y and Z are 3, 11 and 17 respectively. Which two elements will show similar chemical properties? Justify.
Ans. X and Y will show similar chemical properties as these have same valence electrons.
X = 2, (1)
Y = 2, 8, (1)
Short Answer Type Questions-I (2 Marks Each)
1. How it can be proved that the basic structure of the Modern Periodic Table is based on the electronic configuration of atoms of different elements?
Ans. Modern periodic table consists of groups and periods, where number of valence electrons determines the group and number of shells determines the period.
2. Write the name, symbol, and electronic configuration of an element X whose atomic number is 11.
Ans. Name – Sodium
Symbol – Na
Electronic configuration – 2, 8, 1
3. Can the following groups of elements be classified as Dobereiner’s triad?
(a) Na, Si, CI
(b) Be, Mg, Ca
Atomic mass of Be-9; Na-23, Mg-24, Si-28, Cl-35, Ca-40.
Justify your answer in each case.
Ans. (a) Na, Si, Cl – Average of atomic masses of Na and Cl is not equal to the atomic mass of Si. (b) Be, Mg, Ca – The average of atomic masses of Be and Ca is equal to the atomic mass of Mg. Atomic mass of Mg =
Atomic mass of Be + Atomic mass of Ca/2
9+40/2 = 49/2 = 24.5
4. An element ‘X’ has mass number 35 and 18 number of neutrons. Therefore, electronic configuration of X= 2, 8, 7. Write atomic number and electronic configuration of ‘X’. Also write group number, period number and valency of ‘X’.
Ans. Atomic number of X = Mass number of X-Number of neutrons
35 – 18 = 17
Therefore, Electronic configuration of X= 2, 8, 7
Group number = 17
Period number = 3
Valency = 8 – 7 = 1 2
5. The atomic number of an element is 14. Examine if this element will have metallic properties or not. Give reason to justify your answer.
Ans. Atomic number =14
Electronic configuration = 2, 8, 4
Hence, the element is silicon. This element exhibits the properties of both metals and non-metals. Hence, it is semi- metal or metalloid.
6. The electronic configuration of an element is 2, 8, 4. State its: (a) group and period in the Modern Periodic Table. (b) name and write its one physical
Ans.
(a) Group – 14, Period -3
(b) Silicon
Metalloid / poor conductor of electricity
7. The electronic configuration of an element ‘X’ is 2, 8, 6. To which group and period of the modern periodic table does ‘X’ belong. State its valency.
Ans. X: 2, 8, 6
(a) Since ‘X’ has three energy shells and period number of an element is equal to the number of energy shells, X belongs to 3rd period.
(b) X has 6 valence electrons it belongs to group 16.
(c) Valency will be 2. To acquire noble gas configuration, it will gain 2 electrons.
8. Define the following terms? (i) Valency, (ii) Atomic size.
Ans.
- Valency: The combining power or the combining capacity of an atom is called its valency.
- Atomic size: atomic size or atomic radius is the distance between the centre of the nucleus and the outermost shell of an isolated atom.
Short Answer Type Questions-I (3 Marks Each)
1. Study the data of the fillowing three categories A. B and C
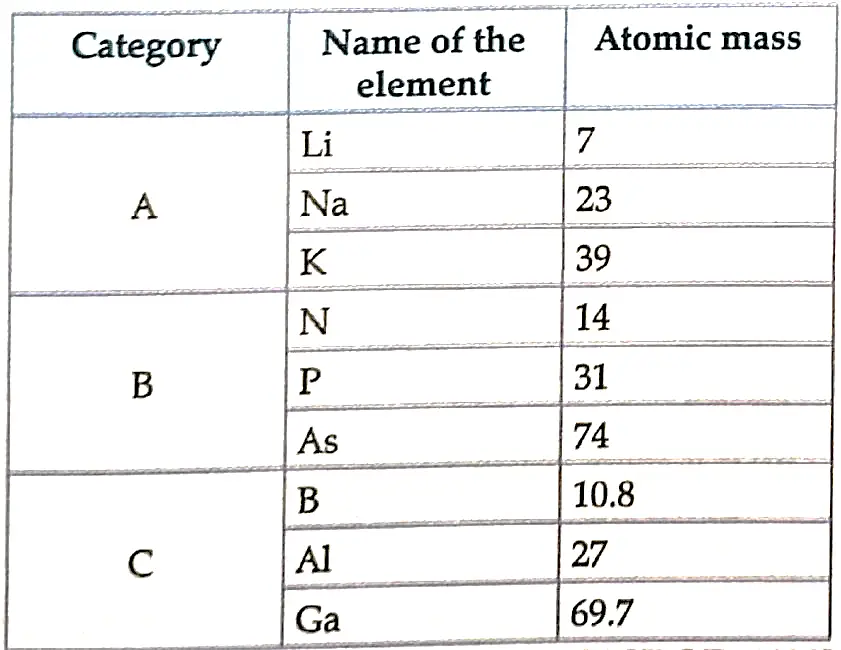
- From the given three categories A, B and C, pick the one which forms Dobereiner’s Triads.
- Why did Mendeleev placed elements of category A, B and C in three different groups?
- Is Newland law of octaves applicable to all the three categories? Give reason to justify your answer.
Ans.
- Category A (Li, Na, K) shows Dobereiner’s triads.
- Mendeleev arranged the elements in increasing order of atomic mass and grouped them as per similar chemical property. The elements in A, B and C are similar among themselves but have different chemical properties.
- No, Newland law of Octaves is not applicable because in the three categories every eighth element will not show same property as first. Also, Newland Octaves law is applicable only upto calcium.
2. Three elements ‘X’, “Y and ‘Z’ have atomic numbers 7, 8 and 9 respectively.
(a) State their positions (Group number and period number both) in the Modern Periodic Table.
(b) Arrange these elements in the decreasing order of their atomic radii.
(c) Write the formula of the compound formed when ‘X’ combines with ‘Z’.
Ans.
(a) X (7) – 2,5 Group 15; Period 2
Y(8) – 2,6 Group 16; Period 2
Z(9) – 2,7 Group 17; Period 2
(b) X > Y > Z
(c) XZ, AI
3. Two elements X and Y have atomic numbers 12 and 16 respectively. To which period of the modern periodic table do these two elements belong? What type of bond will be formed between them and why? Also give the chemical formula of the compound formed.
Ans. Electronic configuration of X: 2, 8, 2, Y: 2, 8, 6
Both X and Y belongs to 3rd period.
Ionic bond will be formed.
Reason: X will lose 2 electrons and Y will gain 2 electrons to complete their octet and become stable.
Formula is XY.
4. The electronic configuration of an element ‘X’ is 2, 8, 6. To which group and period of the modern periodic table does ‘X’ belong. State its valency and justify your answer in each case.
Ans. X: 2, 8, 6
- Since ‘X’ has three energy shells and period number of an element is equal to the number of energy shells. X belongs to 3rd period.
- X has 6 valence electrons it belongs to group 16.
- Valency will be 2. To acquire noble gas configuration, it will gain 2 electrons.
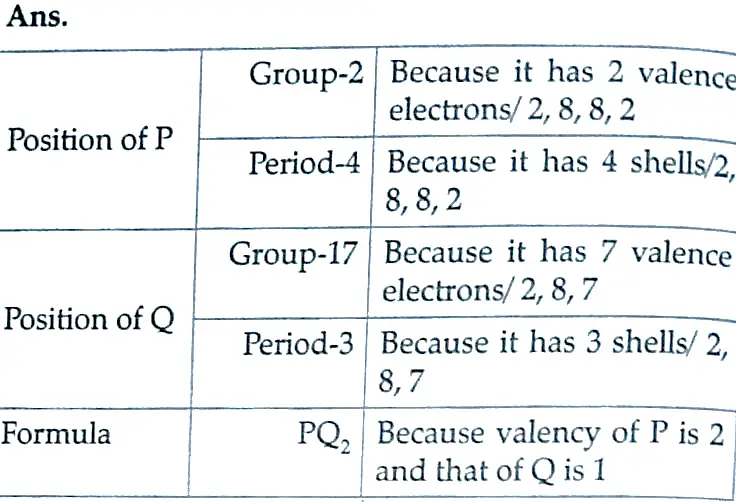
5. Write the electronic configuration of two elements P (atomic number 17) and Q (atomic number 19) and determine their group numbers and period numbers in the Modern Periodic Table.
Ans. Electronic configuration of ‘P- 2, 8, 7
Group number- 17
Period number-3rd
Electronic configuration of ‘Q’-2, 8, 8, 1
Group number 1
Period number – 4th
6. Consider the following elements (atomic numbers are given in parenthesis) Ca (20); K(19); F(9); Be(4)
(a) Select:
(i) The elements having one electron in the outermost shell.
(ii) Two elements of the same group. Write the number of this group.
(b) Write the formula of the compound formed by the union of Ca (20) and the element X(2, 8, 7).
Ans. (a) (i) K (Potassium – 2, 8, 8, 1)
(ii) Be and Ca in same group because both have same number of valence electrons in their outermost shell. The number of this group is 2.
(b) Ca X
Valency 2 1
Thus, the formula of the compound is Ca1X2, CaX2,
7. An element P (atomic number 20) reacts with an element Q (atomic number 17) to form a compound. Answer the following questions by giving reason: Write the position of P and Q in the Modern Periodic Table and the molecular formula of the compound formed when P reacts with Q.
8. The atomic number of an element is 20.
(a) Write its electronic configuration and determine its valency.
(b) Is it a metal or a non-metal?
(c) Write the formula of its chloride.
(d) Is it more reactive or less reactive than Mg (atomic number 12)? Give reason for your answer.
Ans. (a) Electronic Configuration, X (20) -2, 8, 8, 2
Valence electrons- 2
Hence valency is 2
(b) It is a metal
(c) XCL,
(d) It is more reactive than Mg as reactivity increases down the group. Mg- III Period and X20 (Ca)- IV Period.
9. Write the electronic configuration of two elements X and Y whose atomic numbers are 20 and 17 respectively. Write the molecular formula of the compound formed when element X reacts with element Y. Draw electron-dot structure of the product and state the nature of the bond formed between both the elements.
Ans. X (20) – 2, 8, 8,
Y (17) – 2, 8, 7
XY2
![]()
10. An element ‘M’ has atomic number 12.
(i) Write its electronic configuration and valency.
(ii) Is ‘M’ a metal or a non-metal? Give reason in support of your answer.
(iii) Write the formula and nature (acidic/basic) of the oxide of M.
Ans.
(i) Electronic Configuration- 2, 8, 2
Valency-2.
(ii) Metal
There are two electrons in its outermost shell, and it easily loses them to form a positive ion.
(iii)
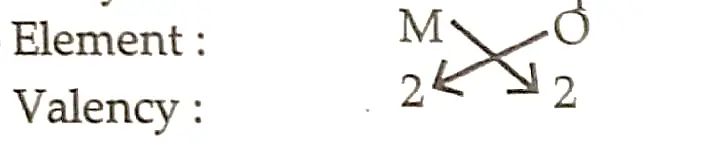
Chemical formula M2O2 = MO
It is a basic oxide.
11. An element’X’ with electronic configuration (2, 8, 2) combines separately with two radicals, (NO3)- and (SO4)2-
(i) Is ‘X’ a metal or a non-metal? Write the nature of its oxide.
(ii) Write the formula of the compounds of ‘X’ formed by the combination of these radicals. Are these compounds covalent or electrovalent?
Ans. (i) X is a metal. Nature of its oxide is basic.
(ii) X(NO3)2, XSO4
These compounds are ionic/electrovalent.
12. State the main aim of classifying elements. Which is more fundamental property of elements that is used in the development of Modern Periodic Table? Name and state the law based on this fundamental property. On which side of the periodic table one can find metals, non-metals and metalloids?
OR
Write the main aim of classifying elements. Name the basic property of elements used in the development of Modern Periodic Table. State the Modern Periodic Law. On which side (part) of the Modern Periodic Table do you find metals, metalloids and non-metals?
Ans.
- Aim of Classification: For systematic and simplified study of elements compounds. and their compounds.
- Basic property: Atomic Number.
- Modern periodic Law: The properties of elements are a periodic function of their atomic number.
- Metals are found on the left side and centre of the Modern Periodic Table.
- Metalloids are found in a zig-zag manner between the metals and the non-metals.
- Non-metals are found on the right side of the Modern Periodic Table.
13. An element ‘X’ belong to 3rd period and group 13 of the Modern Periodic Table.
(a) Determine the valence electrons and the valency of ‘X’.
(b) Molecular formula of the compound formed when ‘X’ reacts with an element ‘Y'(atomic number = 8).
(c) Write the name and formula of the compound formed when ‘X’ combines with chlorine.
Ans. (a) Group 13 means valence electrons are 3 and valency is 3.
(b)
Y (8) – 2, 6 X= 2,8,3
Valency – 2 Valency – 3
Compound formed – X2Y3/Al2O3
(c)
X Chlorine Cl
Valency -3 Valency -1
Compound formed – XCI3 /AICI3
14. The following table shows the position of five elements A,B,C,D and E in the modern periodic table.
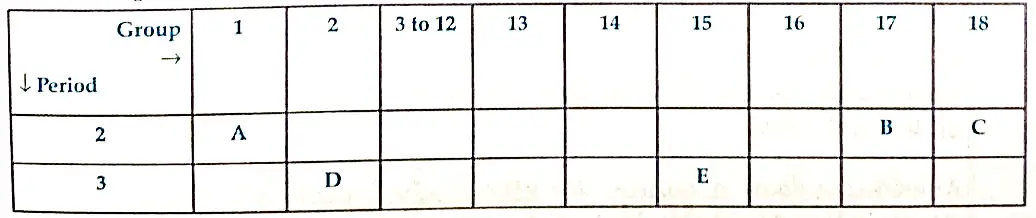
Answer the following giving reasons:
(i) Which elements is a metal with valency two ?
(ii) Which elements is least reactive?
(iii) Out of D and E Which element has a smaller atomic radius ?
Ans. (i) D, as it is on the left side of the table in group 2.
(ii) C, as it is in the group 18/Noble gas.
(iii) E, as we move from left to right across a period, atomic radius decreases.
15. Define atomic size. Give its unit of measurement. In the modern periodic table what trend is observed in the atomic radius in a group and a period and why is it so?
Ans.
- Atomic Size: The distance from centre of nucleus to outermost shell of an atom is atomic radius.
- Doubling the radius defines the atomic size.
- Atomic size is measured in Angstroms, (A°). where 1 Angstroms = 10-10 metres.
- Along the period from left ro right atomic radius decreases.
- Reason: Nuclear charge increase on going down the group.
- Down the group: Atomic radius increases
- Reason: Number of shells increases on going down the group.
16. Define groups in the Modern Periodic Table. How do valency, atomic size and metallic character vary in a group.
Ans. Group: Vertical columns in the modern periodic table are known as ‘groups’. There are 18 groups.
- Valency remains same in a group.
- Atomic size increases from top to bottom in a group.
- Metallic character also increases from top to bottom in a group.
17. How does the atomic radius of the elements change on going
(i) from left to right in a period, and
(ii) down a group in the Modern Periodic Table? Give reason in support of your answer.
Ans.
(i) Atomic radius decreases.
Reason: Nuclear charge increases which tends to pull the electrons closer to the nucleus.
(ii) Atomic radius increases.
Reason: Number of shells increases on going down the group.
18. Write the names given to the vertical columns and horizontal rows in the Modern Periodic Table. How does the metallic character of elements vary on moving down a vertical column? How does the size of atomic radius vary on moving left to right in a horizontal row? Give reason in support of anyour answer in the above two cases.
Ans. Vertical Column-Groups
Horizontal Rows -Periods
(i) Metallic character increases.
Reason: Ability to lose electrons increases on moving down the group due to increase in distance between the nucleus and the valence electrons/ decrease in the attraction between the nucleus and the valence electrons.
(ii) Atomic radius decreases.
Reason: The nuclear charge increases on moving from left to right across a period resulting in increase in the attraction between the nucleus and the valence electrons.
19. Na, Mg and Al are the elements of the 3rd period of the Modern Periodic Table having group number 1, 2 and 13 respectively. Which one of these elements has the ?
(a) highest valency,
(b) largest atomic radius, and
(c) maximum chemical reactivity ?
Justify your answer stating the reason for each.
Ans. (a) highest valency
Na – K L M Mg – K L M Al – K L M
2 8 1 2 8 2 2 8 3
Clearly, highest valency is 3 i.e of aluminum as it can lose its 3 valence electrons to become Al
(b) largest atomic radius → Sodium (na)
Reason → Because atomic Size decreases along a period from left to right since Na is present at most left side, it has more atomic radius.
(c) Maximum chemical Reactivity → Na (Sodium)
Reason → Sice all three are metals chemical Reactivity means ability to lose electrons. Since ability to lose electrons decrease along a period, Na would be most metallic and hence chemically most reactive.
20. What is periodicity in properties of elements with reference to the Modern Periodic Table? Why do all the elements of the same group have similar properties? How does the tendency of elements to gain electrons change as we move from left to right in a period? State the reason of this change.
Ans.
- Repetition of similar properties of elements after regular intervals.
- Because of the same number of valence electrons.
- It increases due to increase in effective nuclear charge which pulls the electrons towards it.
21. Write the number of periods and groups in the Modern Periodic Table. How does the metallic character of elements vary on moving (i) from left to right in a period, and (ii) down a group? Give reason to justify your answer.
Ans.
- Periods -7, Groups – 18
- Metallic character decreases along the period because effective nuclear charge increases on the valence electrons hence decrease in tendency to lose electrons.
- Metallic character increases down a group because effective nuclear charge experienced by valence electrons decrease, hence tendency to lose electron increases.
22. How does the tendency of the elements to lose electrons change in the Modern Periodic Table in (i) a group, (ii) a period and why?
Ans.
(i) Increases down a group.
Reason: At each succeeding element down a group, the number of shells increases, so the distance of the valence shell from the nucleus increases, the effective nuclear force of attraction decreases in the last shell, so it becomes easy for the atom to lose electrons.
(ii) Decreases in a period from left to right.
Reason: As the effective nuclear charge on the valence electron increases, the attraction between the valence electron and nucleus increases, so it becomes difficult to lose electrons.
23.
(i) Name the element with atomic number 17.
(ii) To which period does it belong?
(iii) To which group does it belong?
(iv) Write its electronic configuration.
Ans.
(i) Chlorine
(ii) 3rd period
(iii) 17th
(iv) 2, 8, 7.
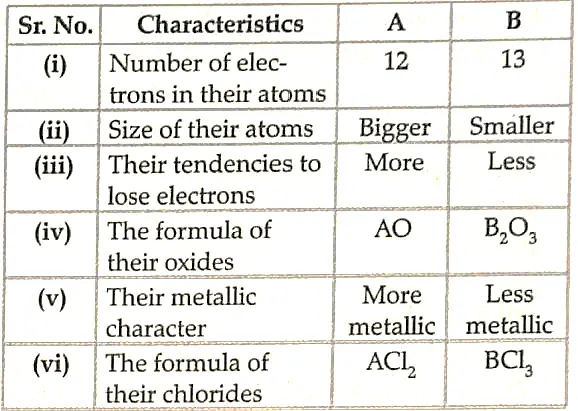
24. Two elements ‘A’ and ‘B’ belong to the 3rd period of Modern periodic table and are in group 2 and 13 respectively. Compare their following characteristics in tabular form:
(i) Number of electrons in their atoms
(ii) Size of their atoms
(iii) Their tendencies to lose electrons
(iv) The formula of their oxides
(v) Their metallic character
(vi) The formula of their chlorides.
Ans.
25. Name any two elements of group one and write their electronic configurations. What similarity do you observe in their electronic configurations? Write the formula of oxide for any of the above said element.
Ans. (i) Two elements of group 1 are Na, K/ Sodium, potassium.
Electronic configurations Na = 2,8,1; K = 2,8,8,1
(ii) Similarity: Both have one valence electron/ One electron in outermost shell.
(iii) Oxide – Na2O/K2O.
26. An element ‘X’ has mass number 35 and number of neutrons 18. Write atomic number and electronic configuration of ‘X’. Also write group number, period number and valency of ‘X’,
Ans. Atomic number of X = Mass number of X- No. of neutrons
= 35 – 18 = 17
Therefore, Electronic configuration of X = 2, 8, 7
Group number =17
Period = 3
Valency = 8-7 = 1
27. The position of eight elements in the modern periodic table is given below where atomic numbers of elements are given in the parenthesis.
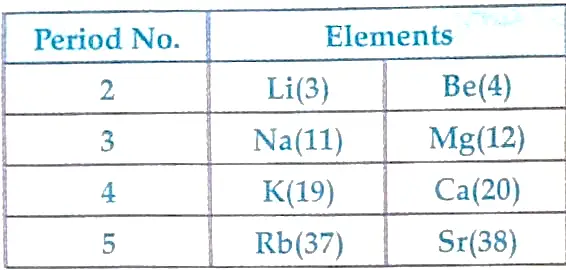
- State electronic configuration of Ca.
- Predict the number of valence electrons in Rb.
- What is the number of shells in Sr?
- Predict whether K is a metal or a non-metal.
- Which one of these elements has the largest atom in size?
- Arrange Be, Ca, Mg and Rb in the increasing order of the size of their respective atoms.
Ans.
- Ca = 2, 8, 8, 2
- Valence electrons in Rb =1
- Five
- Metal
- Rb is biggest in size.
- Be < Mg < Ca < Rb.
28. Four elements A, B, C and D have atomic numbers 12, 13, 14 and 15 respectively.
Answer the following questions giving reasons:
(i) What is the number of valence electrons and valency of D?
(ii) Which of them will have largest atomic radii?
(iii) Which of these elements will form the most basic oxide?
Ans. (i) Valence electrons in ‘D’ = 5 and Valency of ‘D’ = 3.
(ii) ‘A’ will have largest atomic radii because atomic radii decrease across a period from left to right. 1
(iii) ‘A’ will form the most basic oxide as it is most metallic.
29. An element X’ belongs to 3rd period and group 16 of the Modern Periodic Table.
(a) Determine the number of valence electrons and the valency of ‘X’.
(b) Molecular formula of the compound when ‘X’ reacts with hydrogen and write its electron dot structure.
(c) Name the element ‘X’ and state whether it is metallic or non-metallic.
Ans.
(a) Electronic Configuration of X -2, 8, 6
Valence electrons = 6
Valency = 8- 6 = 2
(b) Formula with hydrogen- H2X or H2S
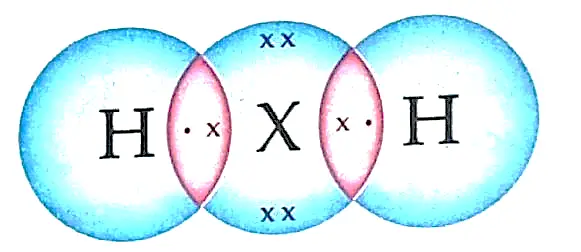
(c) Sulphur; Non-metal
30. An element ‘M’ with electronic configuration (2, 8, 2) combines separately with (NO3), (SO4)2 and (PO4)3– radicals. Write the formula of the three compounds so formed. To which group and period of the Modern Periodic Table does the elements ‘M’ belong? Will ‘M’ form covalent or ionic compounds? Give reason to justify your answer.
Ans.
- The electronic configuration (2, 8, 2) of the element ‘M’ suggests that it belongs to group 2 and period 3 of the Modern Periodic Table and its valency is 2.
- The chemical formula of the compounds are:
- M(NO3)2, Mg (NO3)2; MSO4 / MgSO4 M3 (PO4)2, Mg3(PO4)2
- ‘M’ will form ionic compounds by losing two electrons
31. In the following table, the positions of six elements A, B, C, D, E and F are given as they are in the Modern Periodic Table:
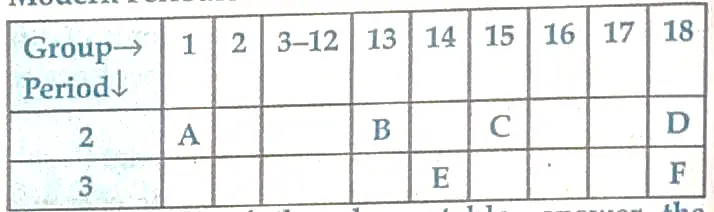
On the basis of the above table, answer the following questions:
(i) Name the element which forms only covalent compounds.
(ii) Name the elem ent which is a metal with valency three.
(iii) Name the element which is a non-metal with valency three.
(iv) Out of B and C, whose atomic radius is bigger and why?
(v) Write the common name for the family to which the elements D and F belong.
Ans. (i) E
(ii) B
(iii) C
(iv) B, because atomic radius decreases from left to right due to increase in the nuclear charge.
(v) Noble gases.
32. The elements 4Be, 12Mg and 20Ca each having two valence electrons in their valence shells are in periods 2, 3 and 4 respectively of the modern periodic table. Answer the following questions associated with these elements, giving reason in each case,
- In which group should they be?
- Which one of them is least reactive?
- Which one of them has the largest atomic size? 1/2 1 12 U[CBSE Term II, O.D. Set-I, 2015]
Ans.
- They all belong to group 2 because all three have 2 electrons in their outermost shell.
- Be is least reactive because it has 2 shells and due to more nuclear change, it is not easy to take electrons from it.
- Ca is the element having largest atomic radius because it has 4 shells. {Hence, it has the largest atomic size.}
Long Answer Type Questions
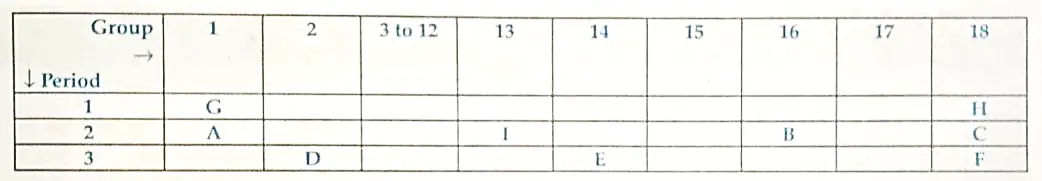
1. The position of certain elements in the Modem Periodic Table are shown below:
Using the above table answer the following questions giving reasons in each case:
- Which element will form only covalent compounds?
- Which element is a non-metal with valency 2?
- Which element is a metal with valency 2?
- Out of H, C and F which has largest atomic size?
- To which family does H, C and F belong?
Ans.
- Element E is Silicon.
- It will form covalent bond only as it has four electrons in its outermost orbit and need only four more electrons to become stable.
- Non-metal with valency 2 is B, which is COxygen.
- Element D is a metal with valency 2.
- Element D is Magnesium. Due to its low electronegativity, it has a higher tendency to donate electrons.
- Element F has the largest atomic size.
- Element F is Argon. Argon occupies 3 energy shells compared to elements H and C, which occupies one and two energy shells.
- Due to this, the atomic radius of Argon is the largest.
- Elements H, C and F belong to Group number 18, which means according to their electronic configuration, their octet is complete and thus these elements are stable.
- They have very low tendency to react with other elements.
- Group 18 elements belong to noble gas family.
2. (a) What was the basis of Mendeleev’s classification of elements?
(b) List two achievements of Mendeleev’s Periodic table.
(c) List any two observations which posed a challenge to Mendeleev’s periodic law.
Ans.
(a) Atomic mass
(b)
- He could classify all the 63 elements known at that time.
- He left gaps for the yet to be discovered elements.
- He predicted the properties of such elements.
(c)
- Position of isotopes
- Irregular increase in atomic masses in going from one element to the next, making the prediction of undiscovered elements difficult.
- Position of Hydrogen.
3.
(a) The modern periodic table has been evolved through the early attempts of Dobereiner, Newland and Mendeleev. List one advantage and one limitation of all the three attempts.
(b) Name the scientist who first of all showed that atomic number of an element is a more fundamental property than its atomic mass.
Ans. (a)
(i) Dobereiner Periodic Table
Advantage: To predict the atomic mass of middle element in each triad.
Limitation: Dobereiner could identify only three triads.
(ii) Newland Periodic table
Advantage: Every eighth element had properties similar to that of first and co-related the properties of elements with their atomic mass.
Limitation: It was only applicable upto Calcium/ only 56 elements and no future element.
(iii) Mendeleev’s Periodic Table
Advantage: Elements with similar properties could be grouped. He predicted the existence of new elements that had not been discovered at that time.
Limitation: No fixed position for hydrogen and isotopes. Atomic masses do not increase in a regular manner.
(b) Henry Moseley: Properties of elements are a periodic function of their atomic number.
4.
(i) Why do we classify elements?
(ii) What are the two criteria used in the development of Modern Periodic Table?
(iii) State the position of (a) metals, (b) non-metals and (c) metalloids in the periodic table.
(iv) Would you place two isotopes of chlorine; Cl-35 and Cl-37 in different slots of the periodic table because of their different atomic masses or in the same slot because their chemical properties are same? Justify your answer.
Ans. (i) To study the properties of elements and to keep the elements with similar properties together.
(ii) Chemical properties of elements and atomic 1 number.
(iii) Metals lie on extreme left; metalloids lie in the middle and non-metals lie on the right side.
(iv) They should be placed in the same slot. Since they have same numbers of electrons/atomic number and Modern Periodic Table is based on atomic number and not on atomic mass.
5(a) List any three observations which posed a challenge to Mendeleev’s Periodic Law.
(b) How does the metallic character of elements vary on moving from
(i) left to right in a period,
(ii) From top to bottom in a period of the Modern Periodic Table ? Given reason for your answer.
Ans. (a) (i) No fixed position of H in the periodic table.
(ii) Position of isotopes not clear.
(iii) Atomic mass does not increase in a regular manner (or any other).
(b) (i) Left to right metallic character decreases. Reason: Effective nuclear charge increases/tendency to loose electrons decrease / electropositivity decreases.
(ii) Top to bottom metallic character increases. Reason: Size of atom increase/tendency to loose electron increases (any one reason).
6. The electrons in the atoms of four elements A, B, C and D are distributed in three shells having 1, 3, 5 and 7 electrons respectively in their outermost shells. Write the group numbers in which these elements are placed in the Modern Periodic Table, configuration of the atoms of B and D and the molecular formula of the compound formed when B andD combine.

Detailed Answer:
Electronic configuration of D: 2, 8, 7
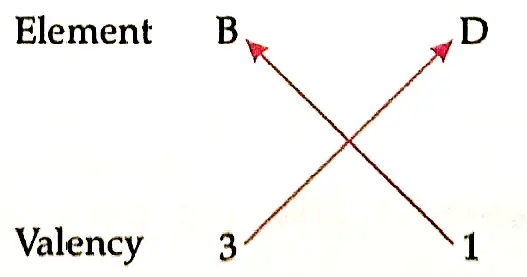
So, the molecular formula will be BD3.
7. Explain giving justification the trends in the following properties of elements, on moving from left to right in a period, in the Modern periodic Table:
(a) Variation of valency.
(b) Change of atomic radius.
(c) Metallic to non-metallic character.
(d) Electronegative character.
(e) Nature of oxides.
Ans. (a) Valency first increases, then decreases
(b) Decreases
(c) Increase
(d) Increases
(e) Change from basic to acidic
Detailed Answer:
(a)
- Valency is the combining capacity of an element.
- Valence electrons are the number of electrons in the outermost shell.
- Valency and valence electrons are same till the number of outermost electrons is 4, but when it goes beyond 4, then the outermost electrons are subtracted from 8 and valency is determined.
- Thus, valency first increases along the period and then decreases.
(b) Along the period, from left to right effective nuclear charge increases as the number of protons increase, due to which force of attraction between nucleus 1 and the valence electron increases therefore, atomic radius decreases.
(c) Along the period, from left to right effective nuclear charge increases as the number of protons increases, due to which force of attraction between nucleus and the valence electron increases thus, it becomes difficult to lose electron across the period and metallic to non-metallic character increases.
(d)
- Along the period, from left to right effective nuclear charge increases as the number of protons increases, due to which force of attraction between nucleus and the valence electron increases.
- Hence the electron from the outermost orbit is difficult to remove, atomic size decreases therefore electronegativity increases.
(e)
- Along the period, from left to right effective nuclear charge increases as the number of protons increases, due to which force of attraction between nucleus and the valence electron increases, hence the electron from the outermost orbit is difficult to 1 remove.
- Therefore, across the period the metallic to non-metallic character increases.
- So, nature of oxide formation across the period changes from basic to acidic.
