Science Term 2 Sample Paper 2022 (Solved)
Class 10 Science Term 2 Sample Paper 2022, (Science) exams are Students are taught thru NCERT books in some of the state board and CBSE Schools. As the chapter involves an end, there is an exercise provided to assist students to prepare for evaluation. Students need to clear up those exercises very well because the questions inside the very last asked from those.
Sometimes, students get stuck inside the exercises and are not able to clear up all of the questions. To assist students, solve all of the questions, and maintain their studies without a doubt, we have provided a step-by-step NCERT Sample Question Papers for the students for all classes. These answers will similarly help students in scoring better marks with the assist of properly illustrated Notes as a way to similarly assist the students and answer the questions right.
Class 10 Science Term 2 Sample Paper 2022
General Instructions:
(i) All questions are compulsory.
(ii) The question paper has three sections and 15 questions.
(iii) Section – A has 7 questions of 2 marks each; Section – B has 6 questions of 3 marks each; and Section – C has 2 case based questions of 4 marks each.
(iv) Internal choices have been provided in some questions. A student has to attempt only one of the alternatives in such questions.
Section – A
1. The molecule of alkene family is represented by the general formula CnH2n. Now answer the following questions: [2]
(a) What do n and 2n signify?
(b) What is the IUPAC name of alkene when n = 4?
2. The atomic number of three elements are given below:

Write the symbol of the element which belongs to (a) group 13, (b) group 15, of the periodic table. State the period of the periodic table to which these elements belong. Give reason to support your answer. [2]
3. The picture given below depicts the process of asexual reproduction in Plasmodium. [2]
(a) Name the process depicted above and define it.
(b) What is meant by asexual reproduction?
4. A student noticed that an organism by mistake was cut into two parts. After sometimes, he noticed that both the parts developed into new individuals. [2]
(a) Name the mode of reproduction used by the organism. State an example of such organism which can multiply by this process.
(b) State the type of cells which can carry out this process.
5. Different species use different strategies to determine sex of a newborn individual. It can be determined by environmental cues or can be genetically determined.
Explain the statement by giving examples for each strategy. [2]
OR
In human beings, the statistical probability of getting either a male or a female child is 50%. Give reasons.
6. Diagram shows lengthwise two sections of a current carrying solenoid. ⨂ indicates current entering into the page, and ⊙ indicates current emerging out of the page. [2]
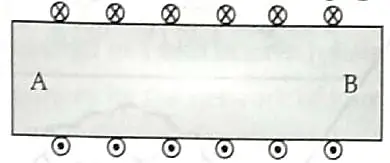
(a) Decide which end of the solenoid A or B will behave as the north pole. Give reason for your answer.
(b) Also draw the field lines inside the solenoid.
OR
A coil of insulated copper wire is connected to a galvanometer as shown in the diagram. State your observations when a bar magnet is: [2]
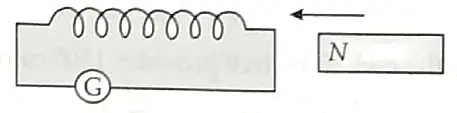
(a) Pushed into the coil with its north pole entering first.
(b) Held at rest inside the coil.
7. In a biology class, teacher taught students about the energy flow by drawing a food chain. In the following food chain, plants provide 5000 J of energy to grasshopper. How much energy will be available to owl from snakes? Can the organisms of any trophic level be removed? [2]
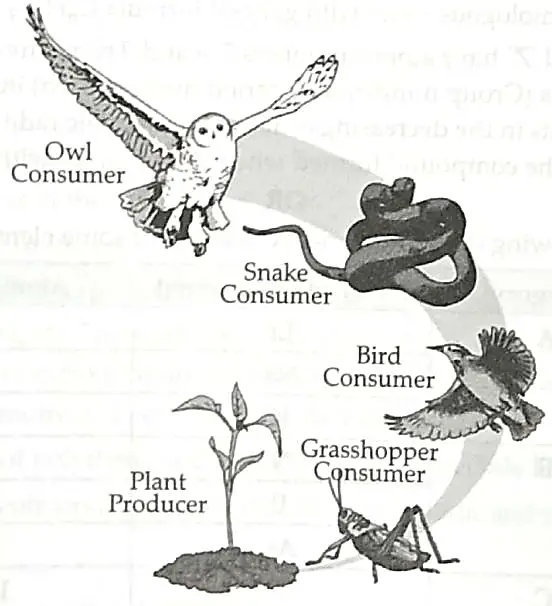
OR
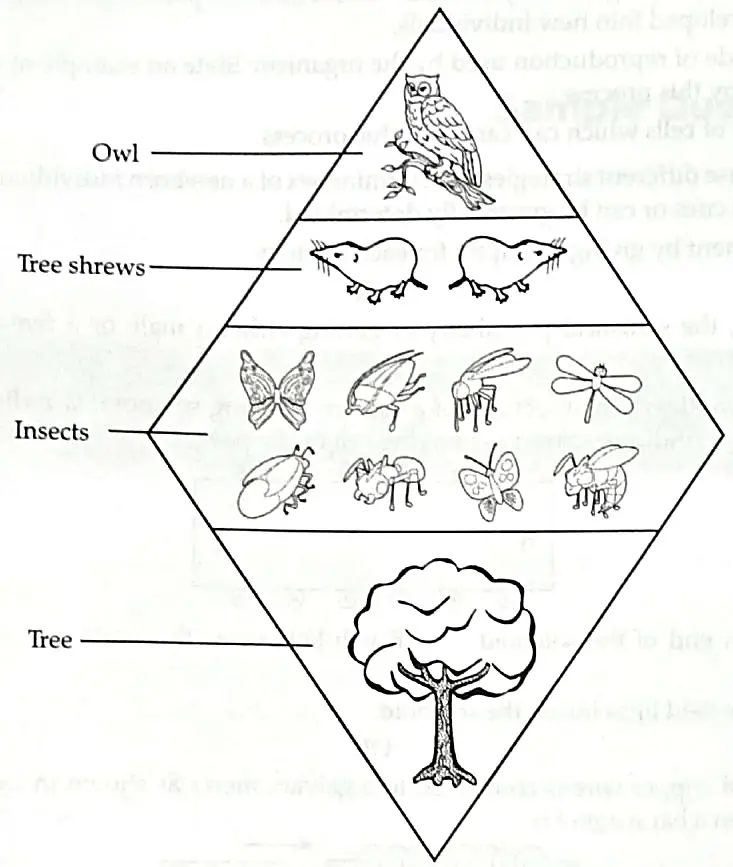
Describe the flow of energy to the owl, if the tree provides 1500 calories of energy to the insects.
Section – B
8. Study the organic compound given in the box. Answer the following questions. [3]
Ethane , Ethene, Ethanoic acid, Ethyne, Ethanol
(a) The compound having functional groups–OH and –COOH.
(b) Gas used in welding.
(c) Homologue of the homologous series with general formula CnH2n+2.
9. Three elements ‘X’, ‘Y’ and ‘Z’ have atomic numbers 7, 8 and 9 respectively. [3]
(a) Identify their positions (Group number and period number both) in the Modern Periodic Table.
(b) Arrange these elements in the decreasing order of their atomic radii.
(c) Write the formula of the compound formed when ‘X’ combines with ‘Z’.
OR
Study the data of the following three categories A, B and C of some elements.
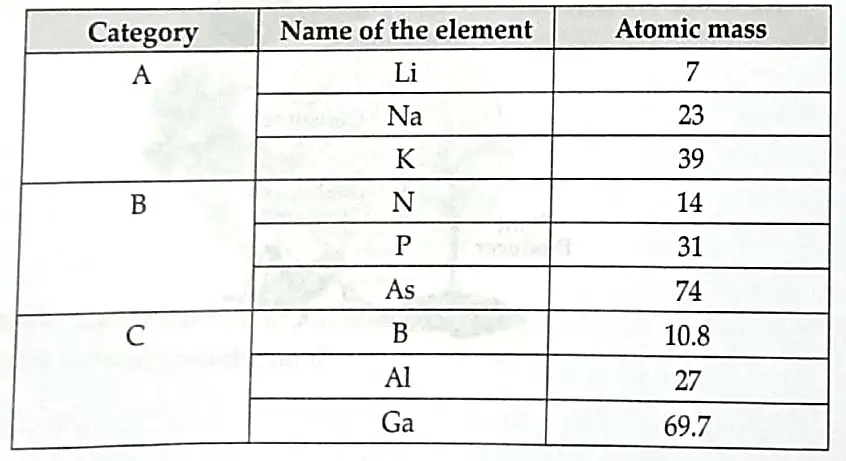
(a) From the given three categories A, B and C, pick the one which forms Dobereiner’s Triads.
(b) Why did Mendeleev placed elements of category A, B and C in three different groups?
(c) Is Newland’s law of Octaves applicable to all the three categories?
Give reason to justify your answer.
10. In one of his experiments with pea plants, Mendel observed that when a pure tall pea plant is crossed with a pure dwarf pea plant, in the first generation F1, only tall plants appear. What happens to the traits of the dwarf plants in this case? When the F1 generation plants were self-fertilised, he observed that, in the plants of second generation F2, both tall plants and dwarf plants were present. Why it happened? Explain briefly. [3]
11. Two coils of resistance R1 = 3Ω and R2 = 9Ω are connected in series across a battery of potential difference 14 V. [3]
(a) Draw the circuit diagram.
(b) Find the electrical energy consumed in 1 min in each resistance.
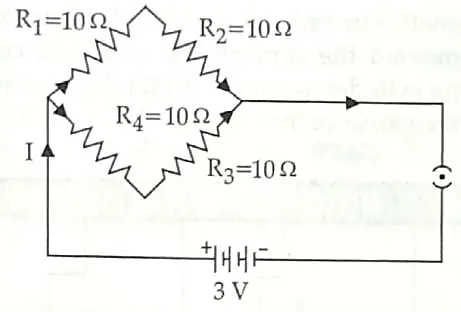
12. Find the current drawn from the battery by the network of four resistors shown in the figure. [3]
OR
For the electric circuit given below, calculate:
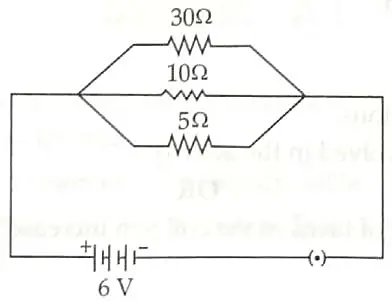
(a) Current in each resistor.
(b) Total current drawn from the battery and
(c) Equivalent resistance of the circuit. [3]
13. While teaching the chapter “Our Environment” the teacher stressed upon the harmful effects of burning of fossil fuels, plastic paper, etc. The students noticed the extensive use of plastic and polythene in daily life can be avoided and the surroundings can be kept clean. They decided to make their school “Plastic and Polythene” free and motivated each other for its minimum use. [3]
(a) Why should the use of polythene and plastic be reduced in daily life?
(b) In what way the students would have avoided the use of plastic and polythene in their school?
Section – C
This section has 02 case-based questions (14 and 15). Each case is followed by 03 sub-questions (a, b and c). Parts a and b are compulsory. However, an internal choice has been provided in part c.
14. Seema crossed pure breed of pea plants having round-yellow seeds with wrinkled green seeds and found that only A-B type of seeds were produced in the F1 generation. When in F1 generation, pea plants having A – B type of seeds were crossbred by self pollination, then in addition to the original round yellow and wrinkled green seeds, two new varieties A – D and C – B types of seeds were also obtained. [4]
(a) What are A -B type of seeds?
(b) State whether A and B are dominant traits or recessive traits.
(c) What are A-D and C-B type of seeds?
OR
Which one will be produced in minimum and maximum number in the F2 generation?
(a) A-B
(b) C-D
15. Asif studied about electromagnetic current induced by changing magnetic field in the class. In the physics laboratory, he experimented the concept. He took two coils of insulated copper wire and wound it over a non conducting cylinder as shown. Coil P had larger number of turns than coil S.. He observed some deflections in the galvanometer and read it. [4]
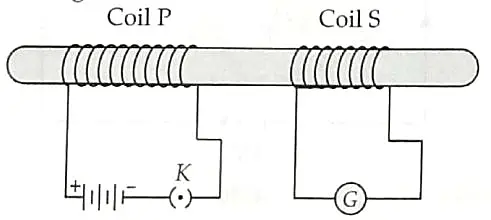
(a) What did he observed when,
(i) key K was closed;
(ii) Key K was opened.
(b) Give reason for his observations.
(c) Define the phenomenon involved in the activity.
OR
What will happen if the number of turns in the coil S in increased?
Solution of Sample Paper
Section – A
1. (a) In general formula CnH2n, n indicates the number of carbon atoms and 2n indicates the number of hydrogen atoms present in the molecule. 1
(b) C4H8, Butene 1
2. (a) A belongs to group 13 because its electronic configuration is 2, 3, i.e., it has 3 valence electrons. 1
(b) ‘B’ belongs to group 15 because its electronic configuration is 2, 5, i.e., it has 5 valence electrons.
They belong to the 2nd period as they both have two shells. 1
3. (a) Multiple Fission. It is a division of single- celled organisms into many daughter cells simultaneously. 1
(b) Creation of a new generation from a single individual or a single parent is known as asexual reproduction. 1
4. (a) The mode of reproduction used by the organism is regeneration. Hydra or Planaria. (any one example) ½ + ½
(b) It is carried out by the specialized cells which can proliferate and make a large number of cells by cell division. 1
5. Environmental Cue:
(i) In some animals, the temperature at which fertilized eggs are kept determines whether the developing animal in the egg is male or female.
(ii) In some animals like snail, individuals can change sex.
Genetic Cue: A child who inherits an X-chromosome from the father will be a girl and one who inherits a Y-chromosome from the father will be a boy. 1 + 1
OR
A child who inherits an “X” chromosome from the father would be a girl (XX) while a child who inherits a “Y” chromosome from the father would be a boy (XY).
Since, the sex of the child is determined by what he/ she inherit from father, therefore the probability of getting either male or female child is 50%. 1 + 1
6. (a) A= North Pole, B = South Pole.
Watching from one end of the solenoid if the current flows anti-clockwise, then North pole is developed at that end and if the current flows clockwise, then South pole is developed at that end. 1
(b)
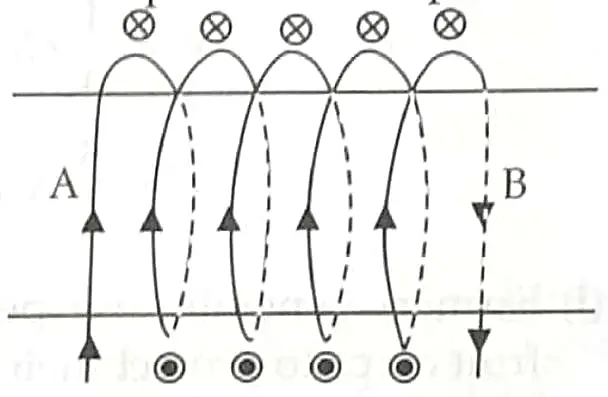 1
1
OR
(a) If a bar magnet is pushed into the coil, an electric current will be induced in the coil due to electromagnetic induction. Therefore, the galvanometer will show some deflection. 1
(b) If a bar magnet is held stationary inside the coil, then no current is induced and therefore there is no deflection in the galvanometer. 1
7. Energy available to owl is 5 J. Energy available to grasshopper = 5000 J.
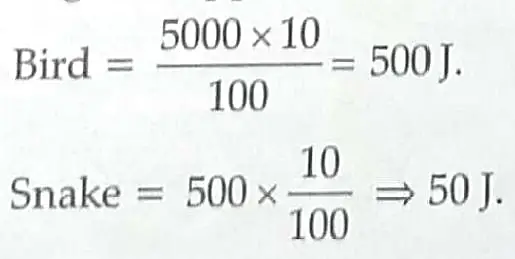
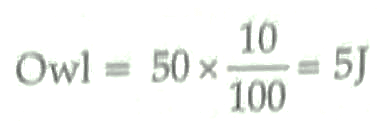
(According to 10 percent Law) ½ + ½
Removal of organisms at a trophic level will upset the whole ecosystem as all categories of organisms are linked through a food chain. The survival of organisms of one trophic level depends on the existence of the members of another trophic level. 1
OR
The insects would provide 10 percent (one tenth) of 1500 calories, or 150 calories, of energy to the shrews. 1
The shrews would provide one tenth of 150 calories, or 15 calories, to the owl. 1
Section – B
8. (a)
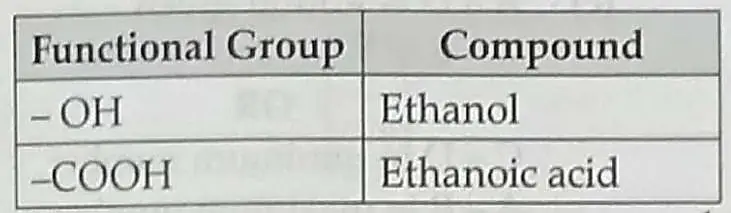 (1)
(1)
(b) Ethyne 1
(c) Alkane : CnH2n+2
Ethane : C2H6 1
9. (a) X (7) – 2, 5. Group 15; Period 2
Y (8) – 2, 6. Group 16; Period 2
Z (9) – 2, 7. Group 17; Period 2 1
(b) X > Y > Z 1
(c) XZ3 1
Detailed Answer:
(a) X = Nitrogen
Y = Oxygen
Z = Fluorine
Groups:
• Nitrogen: Group 15
• Oxygen: Group 16
• Fluorine: Group 17
Period: 2
(b) Across a period, atomic radius decreases from left to right in the modern periodic table. Arrangement of element according atomic radius in decreasing order:
Nitrogen > Oxygen > Fluorine
(c) When X combines with Z, compound formed is XZ3

Thus, Compound formed is XZ3
OR
(a) Category A (Li, Na, K) forms Dobereiner’s triads. 1
(b) Mendeleev arranged the elements in increasing order of atomic mass and grouped them as per similar chemical property. The elements in A, B and C are similar among themselves but have different chemical properties. 1
(c) No, Newland’s law of Octaves is not applicable as it states that the chemical elements are arranged according to increasing atomic masses, those with similar physical and chemical properties occur after each interval of the eighth element. 1
10. The dwarf traits of the plants is not expressed in the presence of the dominant tall trait. 1
In the F2 generation, both the tall and dwarf traits are present in the ratio of 3 : 1, respectively. This showed that the traits for tallness and dwarfness are present in the F1 generation, but the dwarfness, being the recessive trait does not express itself in the presence of tallness i.e., the dominant trait. 2
11. (a) The circuit diagram:
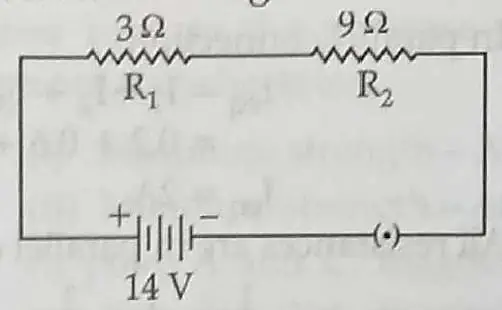
(b) Given: R1 = 3Ω
R2 = 9Ω
In series connections
Rs = R1 + R2
= 9 + 3 = 12Ω
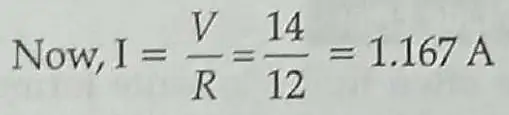
Electric energy consumed in R1
H1 = I2R1t = (1.167)2 x 3 x 60 = 245.14J
Electric energy consumed in R2
H2 = I2R2t = (1.167)2 x 9 x 60 = 735.42J 2
12. Given, V = 3 V
R1 = R2 = R3 = R4 = 10Ω
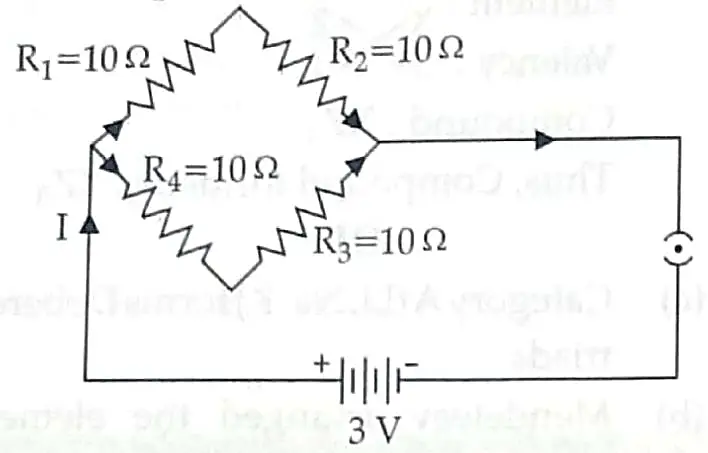
In the given circuit, R1 and R2 are in series. R3 and R4 are also in series connection
R5 = R1 + R2 = 10 + 10 20Ω
R6 = R3 + R4 = 10 + 10 20Ω
Now, R5 and R6 are in parallel connection
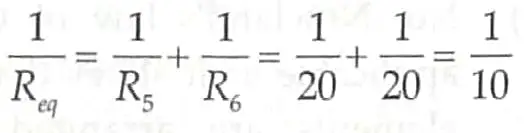
Req = 10 Ω
By Ohm’s law, V= IR
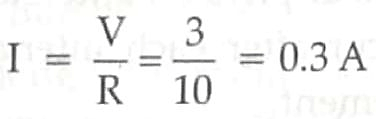 [1 + 1 + 1]
[1 + 1 + 1]
OR
(a) From Ohm’s law,
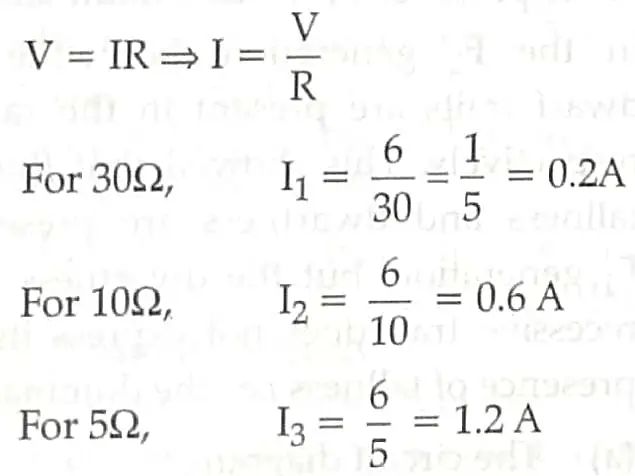
(b) In parallel connection,
Ieq = l1 + I2 + I3
= 0.2 + 0.6 + 1.2
leq = 2A
(c) All resistances are in parallel connection

13. (a) Because they are non-biodegradable. Plastic and polythene cannot be degraded by the activity of microorganisms. 1
(b) (i) Carrying tiffin and water in steel containers.
(ii) Encourage the use of ink pens instead plastic pens. (ball pens are made up of plastic)/or any other (Any two) 1 + 1
Section – C
14. (a) A-B type of seeds-Round in shape and yellow in colour 1
(b) A-B type is dominant traits. 1
(c) A-D = Round-green
C-B = Wrinkled- yellow 1 + 1
OR
C-D in minimum number
A-B in maximum number 1 + 1
15. (a) (i) A momentary deflection was shown by the galvanometer. ½
(ii) A momentary deflection was shown by the galvanometer but in the opposite direction. ½
(b) When key was closed or opened, the current in the coil P changed, therefore the magnetic field linked with coil S will change and hence a current was induced in it. 1
(c) Electromagnetic induction: The process by which a changing magnetic field in a conductor induces current in another conductor. 2
OR
If the number of turns in coil S is increase, the induced current will be more and the deflection of the galvanometer will also be more.